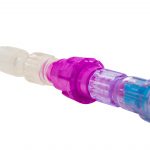
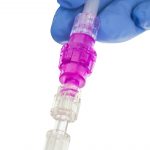
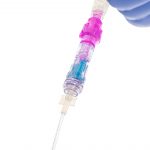
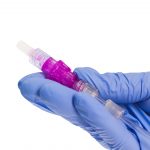
Every Line. Every Time.*
Break away from the expense and pain of IV dislodgements
*Please see product Instructions for Use for a full list of indications and contra-indications.
Pediatric Patients are the most at-risk for vascular access failure.
Two pediatric studies outline IV dislodgement and dislodgement-related failure at 51.9% and 59%, respectively.
- Abdelaziz. B. (2017, Dec 19) Peripheral venous catheter complications in children: predisposing factors in a multicenter prospective cohort study
“The flagship Orchid SRV is a plug and play device that completes a continuum of care for our patients. No new education is required to implement the SRV.”
Largest IV DIslodgement study ever conducted
A study published in J-AVA, conducted by Nancy Moureau, PhD, RN, CRNI, CPUI, VA-BC, and funded by Linear HealthSciences.


1,561 Clinicians surveyed
Over 95% of clinicians surveyed agreed that IV dislodgements continue to pose significant safety risks for patients and hospitals.
PIV, PICC, Midline, & Central Line risks
Over 2/3 of clinicians “often” see dislodgement occurring, and say that 1/2 of catheters still dislodge even with extra securement.


MEDGADGET
medGadget, a leading medical technology publication, highlighted Linear Health Sciences in this article.

MEDTECH INNOVATOR
Selected as a semi-finalist for the Medtech Innovator 2016 competition, Linear Health Sciences received a scholarship to the Virtual Accelerator program.

ANGEL MD
Angel MD, the largest network of medical startups in the world, has singled out Linear Health Sciences as a #FeatureStartup.

2016 INNOVATOR OF THE YEAR
Linear Health Sciences was selected as a finalist for the Journal Record’s 2016 Innovator of the Year award, given to outstanding Oklahoma-based companies.